How to use calcusyn
Chemical–physical data and elemental analyses for 2–13 are reported in Tables S2 and S3 (Supporting Information), respectively high-performance liquid chromatography (HPLC) traces for compounds 7– 10 are reported in Figures S20–S23 (Supporting Information). Diacid 13 was prepared by the hydrolysis of 12 with 2 N potassium hydroxide in ethanol. 12 was prepared by reaction of 1 with ethyl propiolate and tetrabutylammonium fluoride in dried tetrahydrofuran (THF). 10′ was prepared by treatment of 10 with cesium carbonate in aqueous methanol, followed by the alkylation of the resulting cesium salt with methyl iodide in dried DMF. 3– 11 were synthesized by alkylation of 1 with the requisite (cyclo)alkyl-/arylalkyl bromide in the presence of anhydrous potassium carbonate in dried DMF. 2 was obtained by alkylation of commercially available 1 with methyl iodide in dried N, N-dimethylformamide (DMF).
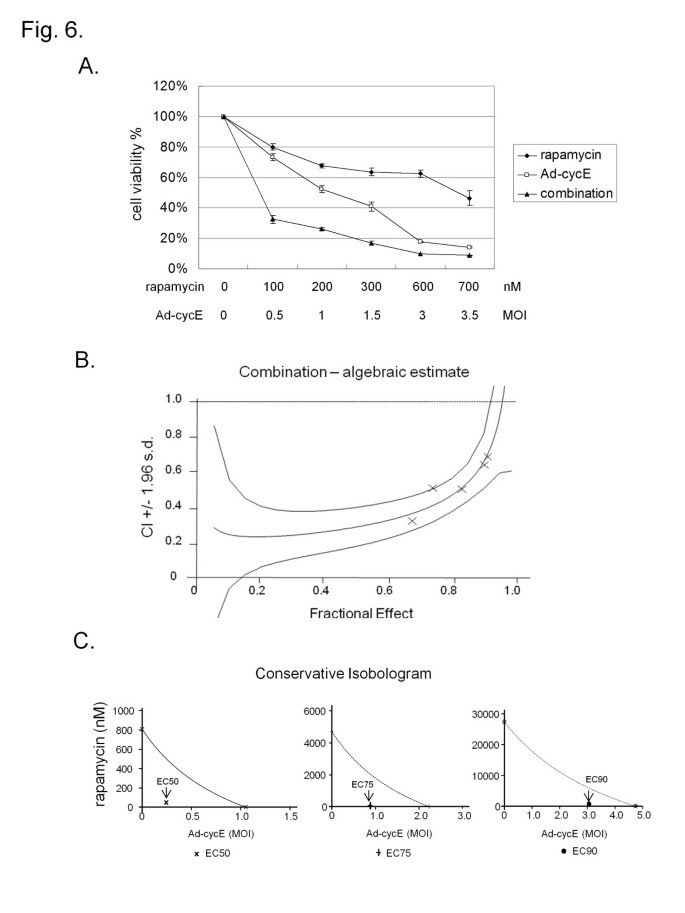
The routes used for the synthesis of 2-substituted-3,4-dihydro-4-oxopyrimidine-5-carboxylic acid derivatives 2–13 are shown in Scheme 1. Thus, given that the prodrug ester forms of these compounds have been widely used in cell biology, it is possible that some of the resultant cellular observations reflect inhibition of MINA53 and/or NO66. The IC 50 values for NOG and 2,4-PDCA are amongst the lowest concentrations reported for the inhibition of human 2OG oxygenases by these two compounds. By contrast, both NOG and 2,4-PDCA, which are reported as relatively broad-spectrum 2OG oxygenase inhibitors, (24) were rather potent inhibitors of both NO66 and MINA53 (NOG IC 50 of 3.5 μM for NO66 and 1.8 μM for MINA53, and 2,4-PDCA IC 50 of 0.11 μM for NO66 and 1.3 μM for MINA53) ( Figure S7). (26) GSK-J1 did not show significant inhibition of MINA53 or NO66 at 100 μM, and IOX-1 was also a poor inhibitor (IC 50 38.5 μM for NO66 and 101.8 μM for MINA53). The assay was validated using known broad-spectrum 2OG oxygenase inhibitors, i.e., pyridine-2,4-dicarboxylate (2,4-PDCA, Figure 3), (24) N-oxalylglycine (NOG, Figure 3), (24) and IOX-1, (25) and the JMJD3/KDM5 inhibitor GSK-J1.
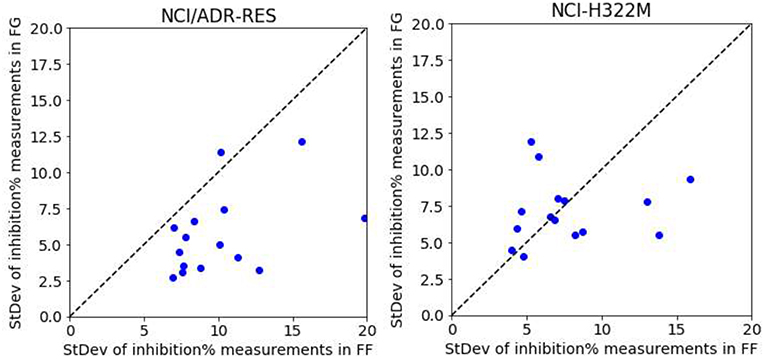
(13) MINA53 is proposed as an important regulator in inflammation and oncology however, the underlying molecular mechanisms by which MINA53 is linked to disease are unclear. (3−11) MINA53 expression is induced by silica particles, suggesting a role for MINA53 in allergen-induced inflammation, (12) and, importantly, in the differentiation of proinflammatory TH17 cells. Elevated MINA53 expression is reported as a poor prognostic indicator, and there is evidence that MINA53 downregulation impairs the proliferation and survival of cancer cells. (2) MINA53 upregulation is linked to solid and hematological tumors, including colon, lung, esophageal, gastric, pancreatic, renal, and hepatocellular carcinomas, breast cancer, leukemias, lymphomas/multiple myelomas, neuroblastomas, and glioblastomas. MYC-induced nuclear antigen (MINA53), also known as mineral dust-induced gene (Mdig) and ribosomal oxygenase 2 (RIOX2), is a JmjC (Jumonji-C) domain-containing 2-oxoglutarate (2OG)-dependent oxygenase localizing to the nucleolus, (1) which is transcriptionally stimulated by the oncoprotein c-MYC. The MINA53 inhibitors show antiproliferative activity with solid cancer lines and sensitize cancer cells to conventional chemotherapy, suggesting that further work investigating their potential in combination therapies is warranted. The MINA53 inhibitors manifest evidence for target engagement and selectivity for MINA53 over KDM4–6. Crystallographic studies with the JmjC demethylase KDM5B revealed active site binding but without direct metal chelation however, molecular modeling investigations indicated that the inhibitors bind to MINA53 by directly interacting with the iron cofactor. These assays enabled the identification of 2-(aryl)alkylthio-3,4-dihydro-4-oxoypyrimidine-5-carboxylic acids as potent MINA53 inhibitors, with selectivity over NO66 and other JmjC oxygenases. Using ribosomal substrate fragments, we developed mass spectrometry assays for MINA53 and the related oxygenase NO66. Despite its anticancer target potential, no small-molecule MINA53 inhibitors are reported. MINA53 is a JmjC domain 2-oxoglutarate-dependent oxygenase that catalyzes ribosomal hydroxylation and is a target of the oncogenic transcription factor c-MYC.
